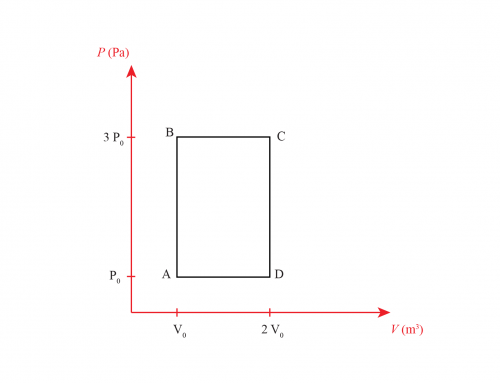
2 moles of an ideal diatomic gas go through a cycle, as seen in the figure. The B-C process is adiabatic.
a) Find pressure, volume, and temperature for the 3 states A, B, and C.
b) Find the change in internal energy, heat, and work carried out by the three processes.
c) Find the total work done by the gas during a cycle.
d) Find the efficiency of this engine.
a) Use the ideal gas law to get the missing states. Be careful with the adiabatic process since the relation from states is slightly modified.
b) Use the definition of work and internal energy to get those variables. Then, use the first thermodynamic’s law to get the heat.
c) Sum all the works done by each process.
d) Use directly the efficiency formula with the values found in b) and c).
a) The ideal gas law states:
\begin{equation*}
PV = nRT,
\end{equation*}
State B: With the given values, solving for \(V_B\) we get:
\begin{equation*}
V_B = 0.022 \, \text{m}^3.
\end{equation*}
State A: The volume is the same as in A. Then:
\begin{equation*}
V_A = 0.022 \, \text{m}^3.
\end{equation*}
Solving for \(T_A\) from the ideal gas law we get:
\begin{equation*}
T_A = 133.3 \, \text{K}.
\end{equation*}
State C: For an adiabatic process:
\begin{equation*}
P_B V_B^{\gamma} = P_C V_C^{\gamma}.
\end{equation*}
Solving for \(V_C\) we get:
\begin{equation*}
V_C = \left(\frac{P_B}{P_C} \right)^{\frac{1}{\gamma}} V_B,
\end{equation*}
or with numerical values:
\begin{equation*}
V_C = 0.049 \, \ \text{m}^3.
\end{equation*}
Solving for \(T_C\) from the ideal gas law we get:
\begin{equation*}
T_C = 292.2 \, \text{K}.
\end{equation*}
b) The work definition is:
\begin{equation*}
W = \int_{V_i}^{V_f} P dV.
\end{equation*}
The change of internal energy is:
\begin{equation*}
\Delta U = \frac{5}{2} nR \Delta T.
\end{equation*}
The first law of thermodynamics states:
\begin{equation*}
\Delta U = Q – W.
\end{equation*}
Using those equation for each process we get:
Process A \(\rightarrow\) B.
\begin{equation*}
\Delta U_{A \rightarrow B} = 11086\, \text{J}.
\end{equation*}
\begin{equation*}
W_{A \rightarrow B} = 0.
\end{equation*}
\begin{equation*}
Q_{A \rightarrow B} = 11086\, \text{J}.
\end{equation*}
Process B \(\rightarrow\) C.
\begin{equation*}
\Delta U_{B \rightarrow C} = -4481.5\, \text{J}.
\end{equation*}
\begin{equation*}
W_{B \rightarrow C} = 4481.5\, \text{J}.
\end{equation*}
\begin{equation*}
Q_{B \rightarrow C} = 0.
\end{equation*}
Process C \(\rightarrow\) A.
\begin{equation*}
\Delta U_{C \rightarrow A} = -6604.5\, \text{J}.
\end{equation*}
\begin{equation*}
W_{C \rightarrow A} = -2700 \, \text{J}.
\end{equation*}
\begin{equation*}
Q_{C \rightarrow A} = -9304\, \text{J}.
\end{equation*}
c) By the sum of the three works found in b) we get:
\begin{equation*}
W_{ \text{Total}} = 1782 \, \text{J}.
\end{equation*}
d) The efficiency can be written as:
\begin{equation*}
\epsilon = \frac{W_{ \text{Total}}}{Q_{\text{abs}}}.
\end{equation*}
The total work was already found. The heat absorbed is \(Q_{A \rightarrow B}\). Then:
\begin{equation*}
\epsilon = 0.16.
\end{equation*}
For a more detailed explanation of any of these steps, click on “Detailed Solution”.
Let \(n = 2\) be the number of moles of gas. Also, let \(P_0 = 10^{5} \ \text{Pa}\) be the pressure at states A and C.
a) Some of the state variables (pressure, volume, and temperature) are already given in the figure’s problem. We can find the rest by using the ideal gas law and the relations between state variables for an adiabatic process.
The ideal gas law states that for n moles of an ideal gas, its pressure \(P\), volume \(V\), and temperature \(T\) are related by the equation:
\begin{equation}
\label{EQ:GAS}
PV = nRT,
\end{equation}
where R is the ideal gas constant. Let’s consider each state separately.
State B: We are given the temperature \(T_B = 400 \ \text{K}\), and the pressure \(P_B = 3 P_0 = 3\cdot 10^5 \) Pa. If we divide eq. \eqref{EQ:GAS} by \(P\), we find that the volume for an ideal gas is given by
\begin{equation}
\label{EQ:V}
V = \frac{nRT}{P},
\end{equation}
and substituting \(T_B\) and \(P_B\) gives
\begin{equation}
\label{EQ:VB}
V_B = \frac{nRT_B}{P_B}
= \frac{nRT_B}{3P_0}
= \left(2 \, \text{mol}\right) \left(8.3145 \, \frac{{\text{J}}}{\text{mol} \, \text{K}}\right)\frac{400 \ \ \text{K}}{3\times10^5 \ \text{Pa}}
= 0.022 \, \text{m}^3.
\end{equation}
State A: Looking at the figure’s problem, we know that \(P_A = P_0 = 10^5 \) Pa. Also, we can notice that \(V_A\) = \(V_B\). Hence, from eq. \eqref{EQ:VB}, we get
\begin{equation}
\label{EQ:VA}
V_A = \frac{nRT_B}{3P_0}
= 0.022 \, \text{m}^3,
\end{equation}
and we can now find the temperature \(T_A\) using the ideal gas law. If we divide eq. \eqref{EQ:GAS} by \(nR\), we obtain that the temperature for an ideal gas is given by
\begin{equation}
\label{EQ:T}
T = \frac{PV}{nR}.
\end{equation}
Substituting eq. \eqref{EQ:VA}, and \(P_A = P_0\) gives
\begin{equation}
\label{EQ:TA}
T_A = P_0 \frac{nRT_B}{3P_0}\frac{1}{nR}
= \frac{T_B}{3}
= \frac{400 \, \text{K}}{ 3} = 133.3 \, \text{K}.
\end{equation}
State C: For this state, we only know that \(P_B = P_0\). Since the process \(B \rightarrow C\) is adiabatic, it is understood that
\begin{equation}
PV^{\gamma} = \text{constant},
\end{equation}
where \(\gamma = \frac{7}{5}\) for a diatomic gas. This relation holds throughout the entire adiabatic process. In particular, if we compare states B and C, we get
\begin{equation}
P_B V_B^{\gamma} = P_C V_C^{\gamma}.
\end{equation}
The only variable we do not know in this equation is \(V_C\), so let’s solve for it. If we divide by \(P_C\), we obtain
\begin{equation}
V_C^{\gamma} = \frac{P_B}{P_C} V_B^{\gamma},
\end{equation}
and if we raise both sides to the \(\frac{1}{\gamma}\) power, we get
\begin{equation}
\left(V_C^{\gamma}\right)^{\frac{1}{\gamma}} = \left(\frac{P_B}{P_C} V_B^{\gamma}\right)^{\frac{1}{\gamma}},
\end{equation}
which we can write equivalently as
\begin{equation}
V_C^{\gamma \frac{1}{\gamma}} = \left(\frac{P_B}{P_C}\right)^{\frac{1}{\gamma}} V_B^{\gamma \frac{1}{\gamma}}.
\end{equation}
Finally, \(\gamma \frac{1}{\gamma} = 1\). Therefore,
\begin{equation}
V_C = \left(\frac{P_B}{P_C} \right)^{\frac{1}{\gamma}} V_B.
\end{equation}
Now, from the figure’s problem \(P_B = 3 P_0, P_C = P_0\). Hence,
\begin{equation}
V_C = \left(3 \frac{P_0 }{P_0} \right)^{\frac{1}{\gamma}} V_B = 3^{\frac{1}{\gamma}} V_B,
\end{equation}
and substituting eq. \eqref{EQ:VB} gives
\begin{equation}
\label{EQ:VC}
V_C = 3^{\frac{1}{\gamma}} \frac{nRT_B }{3P_0}
= 3^{\frac{5}{7}} \left(2\ \text{mol}\right)\left(8.3145\ \frac{\text{J} }{\text{mol} \ \text{K}}\right)\frac{400 \ \text{K}}{3\times10^5 \ \ \text{Pa}}
= 0.049 \ \, \text{m}^3.
\end{equation}
We can now use the ideal gas law to find the temperature. If we divide eq. \eqref{EQ:GAS} by \(nR\), we get
\begin{equation}
T = \frac{PV}{nR},
\end{equation}
In particular, at state C, we have
\begin{equation}
T_C = \frac{P_CV_C}{nR},
\end{equation}
and substituting \(P_C = P_0\) and eq. \eqref{EQ:VC} yields
\begin{equation}
\label{EQ:TC}
T_C = \frac{P_0}{nR} 3^{\frac{1}{\gamma}} \frac{nRT_B }{3P_0} = 3^{\frac{1}{\gamma}} \frac{T_B}{3} = 3^{\frac{1}{\gamma}} 3^{-1} T_B
= 3^{\frac{1}{\gamma} -1} T_B
= 3^{\frac{5}{7} – 1}(400 \, \text{K})
= 292.2 \, \text{K}.
\end{equation}
b) We can find the internal energy of each state directly from the temperatures found in part a by using the equipartition theorem. We can then use these internal energies to find the change in internal energy during each process. Next, we can use the area under the curve of the P-V diagram to find the work done by the gas during each process. Finally, we can use the first law of thermodynamics to find the heat exchanged during each process.
Internal Energy
The equipartition theorem states that for \(n\) moles of a gas in equilibrium, there is an average internal energy of \(\frac{1}{2} nRT\) for each degree of freedom. A diatomic gas has 5 degrees of freedom because for each molecule there is translational Kinetic energy in three dimensions and rotational Kinetic energy over two rotation axes. Therefore, the internal energy \(U\) of \(n\) moles of a diatomic gas is given by
\begin{equation}
\label{EQ:U}
U = 5 \left(\frac{1}{2} nRT\right) = \frac{5}{2} nRT.
\end{equation}
We can substitute the temperatures found in part a in this equation to obtain expressions for the internal energy at all four states. Let’s consider each process separately:
Process A \(\rightarrow\) B: The change in internal energy is defined as
\begin{equation}
\label{EQ:pre_DUAB}
\Delta U_{A \rightarrow B} = U_B – U_A.
\end{equation}
Now, according to eq. \eqref{EQ:U}, the internal energies \(U_B\) and \(U_A\) are given by
\begin{equation}
U_A = \frac{5}{2} nRT_A,
\end{equation}
and
\begin{equation}
U_B = \frac{5}{2} nRT_B.
\end{equation}
Substituting eq. \eqref{EQ:TA} and \(T_B = 400 \ \text{K}\) yields
\begin{equation}
\label{EQ:UA}
U_A = \frac{5}{2} nR \frac{T_B}{3} =\frac{5}{6} nR T_B
= 5543\ \text{J} .
\end{equation}
and
\begin{equation}
\label{EQ:UB}
U_B = \frac{5}{2} nR T_B
= \frac{5}{2} \left(2\ \text{mol}\right)\left(8.3145\ \frac{\text{J} }{\text{mol} \ \text{K}}\right)\left(400 \ \text{K}\right)
= 16629\ \text{J} .
\end{equation}
Therefore, inserting these equations in eq. \eqref{EQ:pre_DUAB} gives
\begin{equation}
\label{EQ:DUAB}
\Delta U_{A \rightarrow B} = 16629 \ \text{J} – 5543\ \text{J}
= 11086\ \text{J} .
\end{equation}
Process B \(\rightarrow\) C: From eq. \eqref{EQ:U}, the internal energy at state C is
\begin{equation}
\label{EQ:UC}
U_C = \frac{5}{2} nRT_C
\end{equation}
and inserting eq. \eqref{EQ:TC}, we get
\begin{equation}
U_C = \frac{5}{2} \left(2\ \text{mol}\right)\left(8.3145\ \frac{\text{J} }{\text{mol} \ \text{K}}\right) \left(292.2 \ \text{K}\right)
= 12147.5 \ \text{J}
\end{equation}
Hence, from this equation and eq. \eqref{EQ:UB}, we obtain that the change in internal energy for this process is
\begin{equation}
\label{EQ:DUBC}
\Delta U_{B\rightarrow C} = U_C – U_B = 12147.5 \ \text{J} – 16629\ \text{J}
= -4481.5\ \text{J} .
\end{equation}
Process C \(\rightarrow\) A: From eqs. \eqref{EQ:UA} and \eqref{EQ:UC}, we get
\begin{equation}
\label{EQ:DUCA}
\Delta U_{C\rightarrow A} = U_A – U_C = 5543 \ \text{J} – 12147.5\ \text{J}
= -6604.5\ \text{J} .
\end{equation}
Work
Now let’s calculate the work done during each process. The work performed by the gas W during a process can be shown to be equal to the area under the curve of the line describing the process in a P-V diagram. This can be formally written as
\begin{equation}
\label{EQ:W}
W = \int_{V_i}^{V_f} P dV.
\end{equation}
Let’s consider each process separately.
Process A \(\rightarrow\) B: The area under the curve described by this process is zero (i.e. the volume does not change during this process), as can be seen in figure 1.
Figure 1: Thermodynamic process for the gas from point A to B. The pressure increases while the volume remains constant; hence, the area under the curve is zero, which means the work done by the gas is also zero.
Hence,
\begin{equation}
\label{EQ:WAB}
W_{A \rightarrow B} = 0.
\end{equation}
These results can be also obtained from eq. \eqref{EQ:W} because both limits of integration are equal and thus the integral is zero.
Process B \(\rightarrow\) C: Since this process is adiabatic, we have \(Q_{B\rightarrow C} = 0\). We can use this fact together with the first law of thermodynamics to find the work done by the gas.
Figure 2: Thermodynamics process of the gas from point B to C. The process is adiabatic, the volume increases while the pressure decreases. The shaded region indicates the area under the curve, which is equivalent to the work done by the gas from B to C. The work done is positive because the volume increases.
The first law of thermodynamics states that for a substance undergoing a thermodynamic process, the change in internal energy equals the heat \(Q\) absorbed by the substance minus the work \(W\) that it does. This can be written as
\begin{equation}
\label{EQ:1LT}
\Delta U = Q – W,
\end{equation}
where a positive sign for \( Q\) indicates that the substance absorbed heat, and a negative sign indicates that it radiated heat. Similarly, when \(W > 0\), the substance did work during the process, and when \(W < 0\), work was done on the substance. Solving for \(W\) yields \begin{equation} W = Q – \Delta U, \end{equation} and for this particular process this eq. can be written as \begin{equation} W_{B \rightarrow C} = Q_{B\rightarrow C} – \Delta U_{B\rightarrow C}. \end{equation} Now, as mentioned above, \(Q_{B\rightarrow C} = 0\), and thus \begin{equation} W_{B \rightarrow C} = – \Delta U_{B\rightarrow C}. \end{equation} Substituting eq. \eqref{EQ:DUBC} gives \begin{equation} \label{EQ:WBC} W_{B \rightarrow C} = -(-4481.5\ \text{J} ) = 4481.5\ \text{J} . \end{equation} As expected, since there is no heat being transferred, all the internal energy lost by the gas is used to do work as it expands.
Process C \(\rightarrow\) A: During this process, the volume changes from \(V_i = V_C\) to \(V_f = V_A\) at a constant pressure of \(P_0\).
Figure 3: Thermodynamics process of the gas from point C to A. The process is isobaric, the volume decreases while the pressure is kept constant. The shaded region indicates the area under the curve, which is equivalent to the work done by the gas from C to A. The work done is negative because the volume decreases.
Hence, from eq. \eqref{EQ:W} we get \begin{equation} W_{C \rightarrow A} = \int_{V_C}^{V_A} P_0 dV = P_0 \int_{V_C}^{V_A} dV = P_0 V|_{V_C}^{V_A} = P_0 (V_A – V_C), \end{equation} and substituting \(P_0 = 10^5 \ \ \text{Pa}\) in eq. \eqref{EQ:VA}, and eq. \eqref{EQ:VC} yields \begin{equation} \label{EQ:WCA} W_{C \rightarrow A} = (10^5 \ \ \text{Pa}) ((0.022 \ \text{m}^3) – (0.049 \ \text{m}^3)) = -2700\ \text{J} . \end{equation}
Heat
We should now find the heat exchanged during the three processes. Since we have already found the work and the change in internal energy of all processes, we can easily find the heat exchanged by using the first law of thermodynamics. Solving for \(Q\) in eq. \eqref{EQ:1LT} yields \begin{equation} \label{EQ:1LT_Q} Q = \Delta U + W. \end{equation} Let’s apply this equation to each process.
Process A \(\rightarrow\) B: Substituting eqs. \eqref{EQ:DUAB} and \eqref{EQ:WAB} in eq. \eqref{EQ:1LT_Q} gives \begin{equation} \label{EQ:QAB} Q_{A\rightarrow B} = 11086 \ \text{J} + 0 = 11086\ \text{J} . \end{equation}
Process B \(\rightarrow\) C: This process is adiabatic. Hence, \begin{equation} \label{EQ:QBC} Q_{B\rightarrow C} = 0. \end{equation}
Process C \(\rightarrow\) A: Substituting eqs. \eqref{EQ:DUCA} and \eqref{EQ:WCA} in eq. \eqref{EQ:1LT_Q} gives \begin{equation} \label{EQ:QCA} Q_{C\rightarrow A} = -6604 \ \text{J} + (-2700\ \text{J} ) = -9304\ \text{J} . \end{equation}
c) The total work W performed by the gas during a cycle is simply the sum of the works performed by the gas during each process. This can be written as \begin{equation} W_{ \text{Total}} = W_{A\rightarrow B} + W_{B\rightarrow C} + W_{C\rightarrow A}, \end{equation} and substituting eqs. \eqref{EQ:WAB}, \eqref{EQ:WBC}, and \eqref{EQ:WCA} gives \begin{equation} \label{EQ:WTOTAL} W = 0 + 4482 \ \text{J} + (-2700 \ \text{J}) = 1782\ \text{J} . \end{equation}
d) The efficiency \(\epsilon\) of an engine performing a thermodynamic cycle is defined as the net work performed \(W\) divided by the heat absorbed by the gas \(Q_{\text{abs}}\). This can be written as: \begin{equation} \label{EQ:EFF} \epsilon = \frac{W}{Q_{\text{abs}}}. \end{equation} In this case, W is given by eq. \eqref{EQ:WTOTAL}. In order to find \(Q_{\text{abs}}\), we should identify at which processes the gas absorbed heat, and sum up the amount of heat absorbed at each of these processes. From eqs. \eqref{EQ:QAB}, \eqref{EQ:QBC} and \eqref{EQ:QCA}, we can notice that \(Q_{A\rightarrow B} > 0, Q_{B\rightarrow C} = 0\) and \(Q_{C \rightarrow A} < 0\). As explained in part (b), we define our sign conventions so that \(Q>0\) indicates that the gas absorbed heat. Therefore,
\begin{equation}
Q_{\text{abs}} = Q_{A\rightarrow B},
\end{equation}
and inserting eq. \eqref{EQ:QAB} gives
\begin{equation}
Q_{\text{abs}} = 11086\ \text{J} .
\end{equation}
Hence, substituting this equation and eq. \eqref{EQ:WTOTAL} in eq. \eqref{EQ:EFF} yields
\begin{equation}
\epsilon = \frac{W_{ \text{Total}}}{Q_{\text{abs}}} = \frac{1782\ \text{J}}{11086\ \text{J}}
= 0.16 = 16.1 \%.
\end{equation}




Leave A Comment